Astatine By: Claire Svihra (as-tuh-teen)
Astatine's History
- Discovered in California
- By Dale Carson and Mackenzie
- In 1940
- Astatine's origine is greek
- "Astatos"
Symbol, Atomic number, Atomic mass
- Astatine's symbol = At
- Atomic number = 85
- Atomic mass = (210)
Melting and Boiling point
- Melting point = 302.0°C
- Boiling point = 337.0°C
Properties
- Solid and low in density
- Non-magnetic
- Halogen
- Unable to conduct heat
- Highly reactive
Appearance
- In some cases shiny
- Almost like a moldy rock
Astatine's Uses
- Astatine has one use
- Cancer treatment
Does Astatine have similarities?
- Astatine is very similar to iodine
- They behave and react similar
- They both seem to gather in the thyroid
- Iodine = element (53, 126.90447)
How is astatine created
- Astatine is smashed with Bismuth and energetic alphaparticles
- Bismuth = a element (83, 208.98038,)
- energetic alpha particles = a particle with 2 neutrons and 2 protons
- Astatine is extremely rare
- Highly reactive
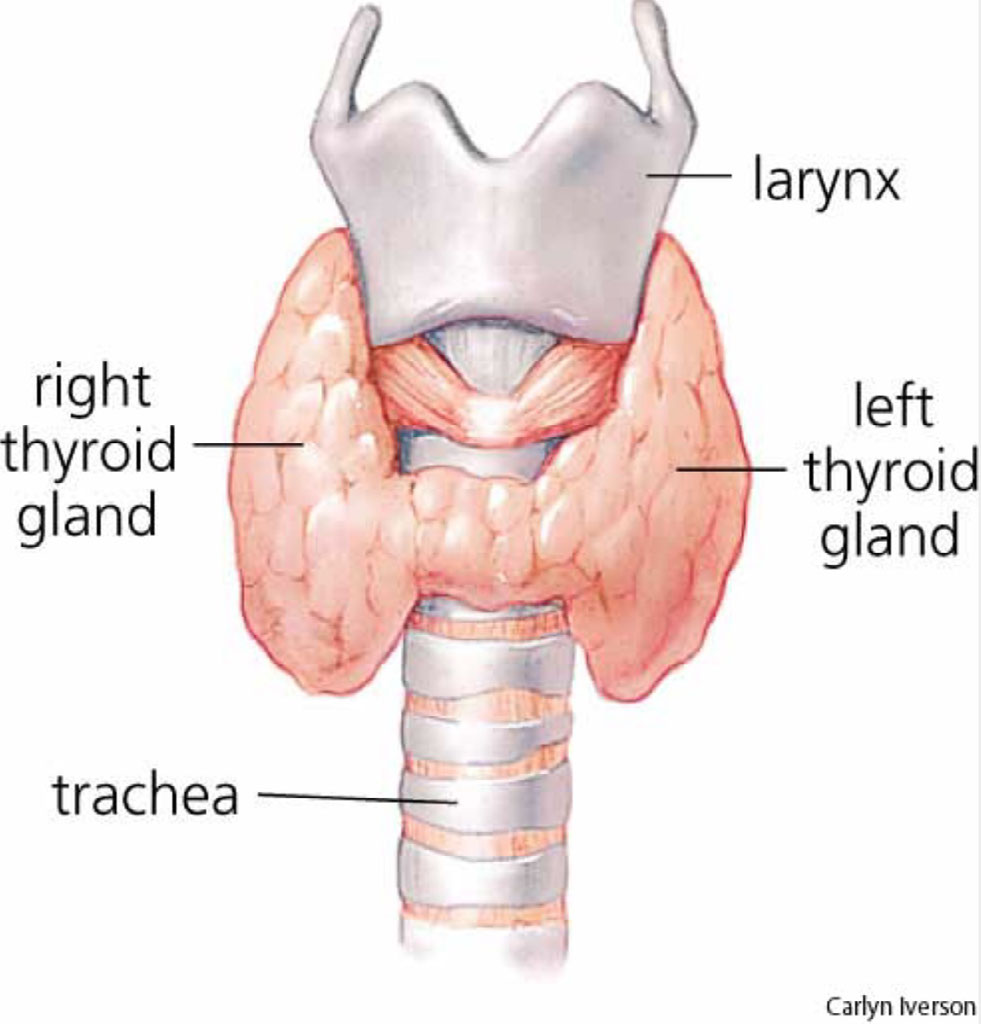
- Is thougth to gather in the thyroid gland
- Which is located in the neck and produces hormones
- Scientist believe there is less than an ounce in the world
Citations
Facts About Astatine." LiveScience. TechMedia Network, 9 Sept. 2013. Web. 19 May 2016.
Bentor, Yinon. "Chemical Elements.com - Astatine (At)." Chemical Elements.com - Astatine (At). Yinon Bentor, 2012. Web. 18 May 2016.
Gagnon, Steve. "The Element Astatine." It's Elemental -. Steve Gagnon, n.d. Web. 19 May 2016.
Freeman, David. "Rarest Element, Astatine, Has Key Property Measured For First Time." The Huffington Post. TheHuffingtonPost.com, 2016. Web. 19 May 2016.
Winter, Mark. "Astatine: The Essentials." Astatine»the Essentials [WebElements Periodic Table]. Mark Winter, 2016. Web. 19 May 2016.
The Periodic Table." Periodic Table of Elements and Chemistry. Chemicool, 2016. Web. 19 May 2016.
Busby, Bruce. "Alpha." Alpha. Bruce Busby, 16 Mar. 2011. Web. 21 May 2016.